A major new review paper titled, High-dose vitamin C (HDVC): A promising anti-tumor agent, insight from mechanisms, clinical research, and challenges, analyzed 150+ studies and found that when vitamin C given by IV Infusion reaches (20,000–30,000 microM/liter concentration in the blood, it becomes a targeted cytotoxic cancer therapy — something past trials missed due to under-dosing.
Historical Perspective
Chemist Linus Pauling championed the potential for huge amounts of IV Vvitamin C to kill cancer cells in the 1970s. His research found that when cancer patients received high doses of IV Vitamin C, they lived four times longer than those who didn’t get the treatment.
However, when Mayo Clinic researchers attempted to recreate Pauling’s findings, they used relatively low dose oral Vitamin C and concluded that Vitamin C offered no benefit in cancer patients’ symptoms or survival rates. Unfortunately, the intentional or not intentional idea of doing similar Clinical Trials with low dose oral Vitamin C was bound to fail and it did. Consequently, due to the failed Mayo Clinic studies, this powerful, cancer-killing agent was essentially forgotten for 30 years.
That is until National Institutes of Health scientist Mark Levine uncovered a key flaw in the The Mayo Clinic research; they only gave their cancer patients low dose oral Vitamin C.
“No matter how much vitamin C you take orally, you can’t get anywhere near enough of the massive doses needed to kill cancer cells,’ according to Dr. Joseph Cullen, MD, Professor Emeritus, University of Iowa, Department of Surgery.
“At low levels, when you take oral vitamin C, it acts as an antioxidant, but when we give it at high levels intravenously, it acts as a pro-oxidant,” Cullen explains.
It is precisely this pro-oxidant effect that kills cancer cells. Dr. Cullen explains that high dose Vitamin C given intravenously generates highly reactive Hydrogen Peroxide (H2O2) that is selectively cytotoxic to cancer compared to normal cells. Cancer cells tend to absorb high levels of Vitamin C.
High-dose intravenous (IV) vitamin C is converted into Dehydroascorbate (DHA) and further into highly reactive hydrogen peroxide H2O2 within the plasma and extracellular fluid that is selectively cytotoxic to cancer cells but is converted back to its healthy anti-oxidant form known as Ascorbate in normal cells.
This process is generally only achievable with pharmacological, high-dose IV administration (75-100 grams per IV infusion) which results in levels of ascorbate in the blood—100 to 500 times higher than that achieved by oral ingestion
The process also makes cancerous cells more susceptible to chemotherapy and radiation, Dr. Cullen says. Normal cells, meanwhile, are unharmed.
“For a therapy this safe, inexpensive, and mechanistically potent, the findings are striking.”
Vitamin C becomes a Cancer Cell killer at high plasma concentrations
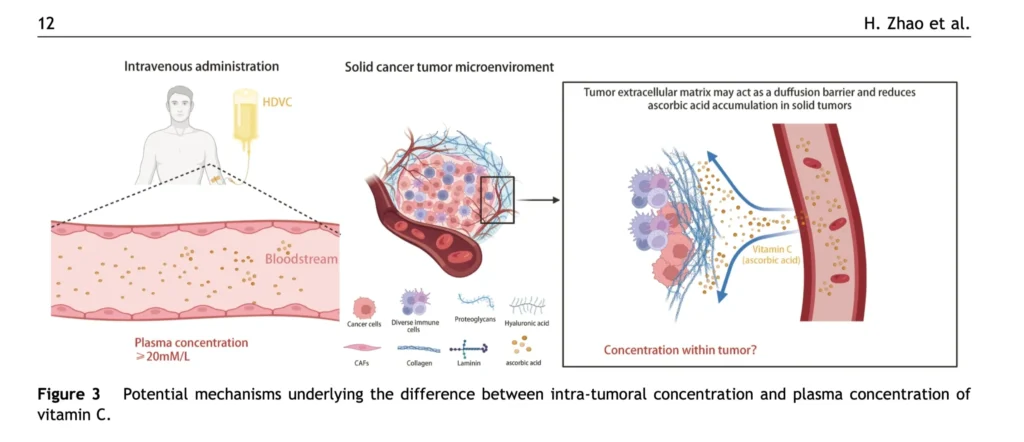
When 75 grams of Vitamin C (Ascorbic Acid) is given by IV infusion, the plasma levels can reach 20,000–30,000 microM/Liter. The highest maximum blood plasma level achievable from oral Vitamin C is 220microM/Liter. The High Dose IV Vitamin C concentration converts Ascorbic Acid into Dehydroascorbic acid (DHA), which generates hydrogen peroxide and hydroxyl radicals inside tumors — but is harmless to normal tissues.
Vitamin C Suppresses the Master Switch of Tumor Survival
Vitamin C is a required cofactor for the enzymes that degrades a central regulator of tumor aggressiveness. High Dose Vitamin C plasma concentrations equal to 20,000-30,000 microM/L are shutting down Cancer Cell Survival, by shutting down:
- angiogenesis
- metastatic signaling
- hypoxia tolerance
- Glucose upregulation into Cancer Cells and replacing it with Vitamin C
Very few other agents directly dismantle this survival pathway.
Vitamin C enhances anti-tumor immunity
High-dose intravenous vitamin C also strengthens the immune system’s ability to attack cancer. It has been shown to:
- Increase T-Helper Cells and Cytotoxic T-Cells infiltration into tumors
- Boost Immunity: IL-12 and enzymes that enhance Cytotoxic T-Cells and Natural Killer Cells
- Upregulate interferon-gamma induced chemokines
- Unleash the body’s immune system to fight cancer by blocking checkpoint proteins
- Enhance T-cell function and proliferation
- Improve natural killer (NK) cell cytotoxicity
- Activate dendritic cells, strengthening antigen/cancer presentation
Together, these amazing natural anti-tumor immune actions are amplified by High Dose Vitamin C (HDVC)
Colorectal, Pancreatic and Lung Cancers are especially sensitive to High Dose Vitamin C Infusions
A Phase III clinical trial even reported significantly improved survival in colorectal cancer patients when high-dose intravenous vitamin C was added to standard therapy.
Early Clinical Trials show Meaningful Survival Improvements
The review summarizes multiple Phase I/II trials where high-dose intravenous vitamin C strengthened standard therapy:
- Pancreatic cancer: major tumor shrinkage in 8 of 9 patients
- Brain Cancer Glioblastoma: overall survival increased from 14.6 → 19.6 months
- Lung cancer: response rates roughly doubled
- Ovarian cancer: reduced chemotherapy toxicity + longer progression-free survival
Across all studies, safety and tolerability were excellent.
High Dose Vitamin C (HDVC) Dosing
The review identifies the dosing regimen required to achieve tumor-selective, cytotoxic plasma levels: 75–100 grams IV per infusion, or >1.0 g/kg IV per infusion, Given 2–3 times per week for 6-8 cycles.
This reliably produces ≥20,000 microM plasma concentrations — the range associated with selective cancer cell killing — while remaining well-tolerated.
The authors also emphasize that most patients in clinical trials never reached the maximum tolerated dose, suggesting the therapeutic ceiling is likely far higher than what past studies explored.