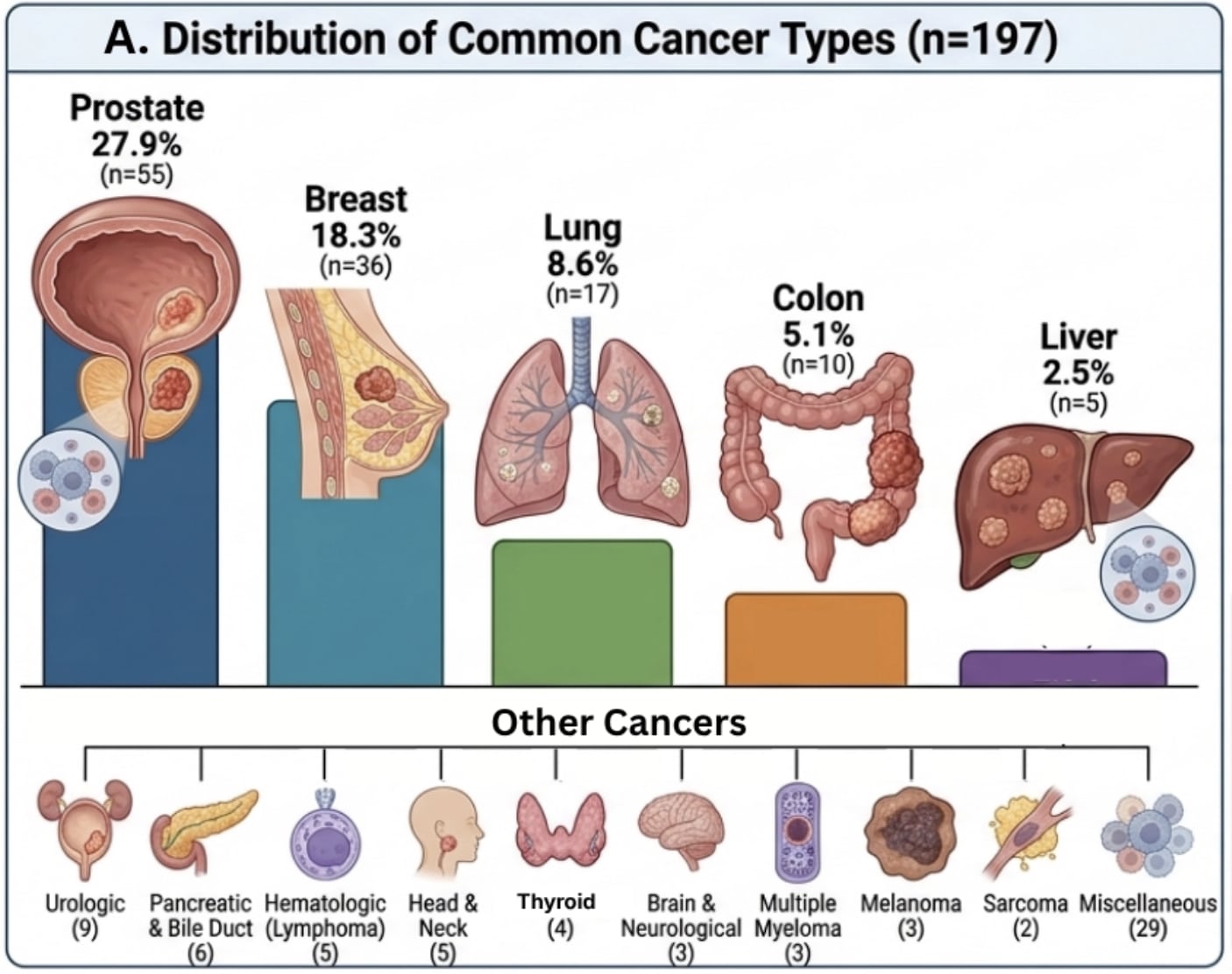
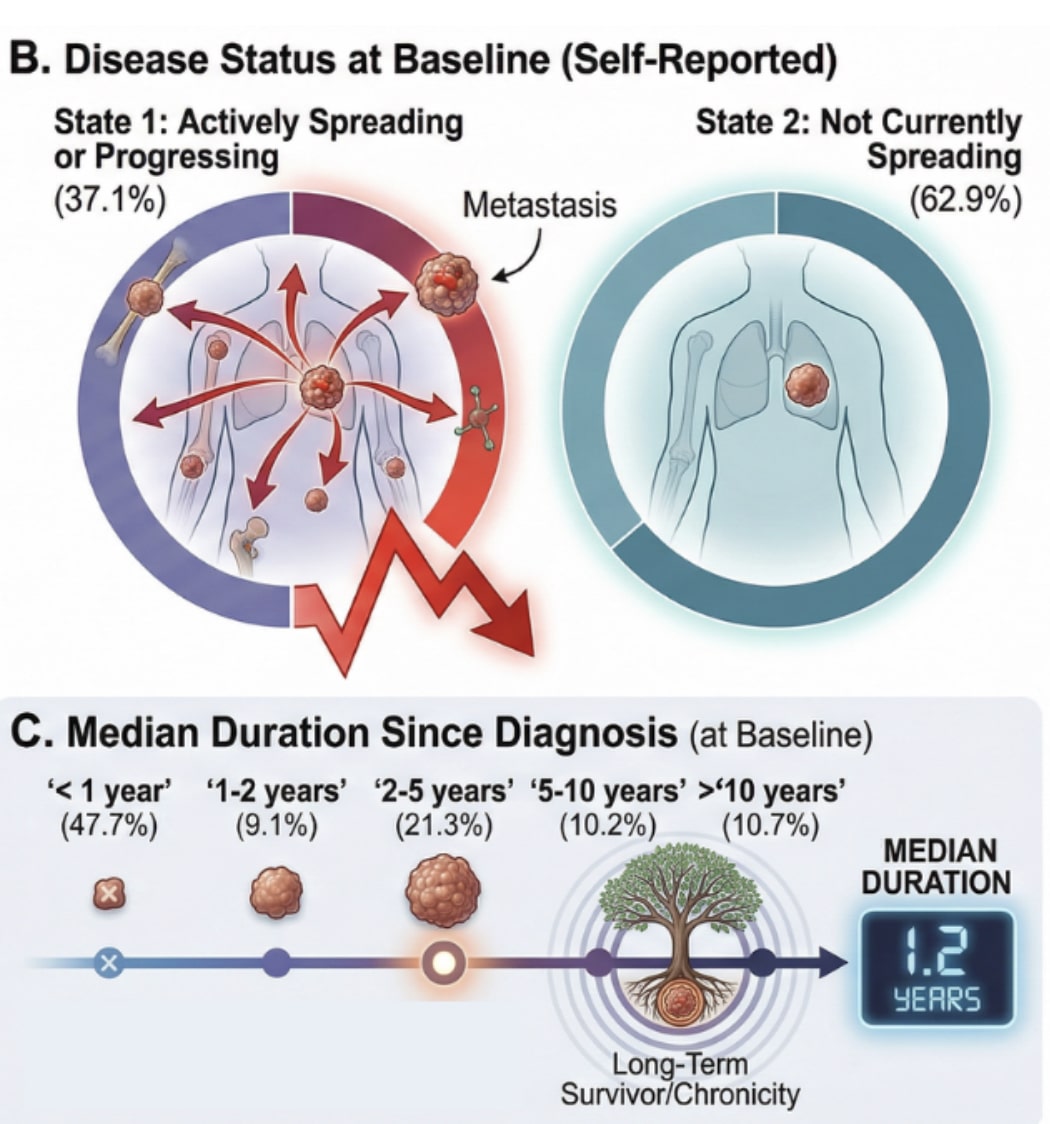
FDA initiating new regulations to limit Compounding Pharmacies and Bio-Identical Hormones
Big Drug Companies want to limit your access to natural, Bio-Identical Hormones, so that the only options you have left are the drugs that only they make, i.e. the synthetic copies of your hormones that they can patent and sell for billions of dollars annually.
These synthetic drugs, like Premarin, Prempro, Estrace, Vivelle-Dot, etc, don’t work as well as Bio-Identical Hormones and cause significant side-effects that are not found in Compounded Bio-Identical Hormones. The only reason Big Drug Companies make the synthetic drugs instead of Bio-Identical Hormones is because they can’t patent something natural that already exists in nature, like Bio-Identical Hormones. God holds the patent to natural Bioidentical hormones. So, in order get a patent and the 20 year exclusivity that comes with it, the drug companies must alter the natural chemical structure of the hormone in order to make it unique enough to claim patent rights.
Before the new FDA regulatory changes, I was able to order Bio-Identical Hormones in bulk from Compounding Pharmacies and dispense them to all of my patients directly, in any amount needed for each individual patient. I had approximately 100 different combinations of the natural Bio-Identical Hormones, including combinations of Estriol, Progesterone and Testosterone, which I dispensed based on the individual symptoms and lab test results of each patient.
The new FDA regulation requires that a prescription for each Bio-Identical Hormone must be written and shipped to each individual patient. This is extremely time consuming and has made it impossible for the Compounding Pharmacy to make all of the unique individual formulas that I typically prescribe for my patients. There simply is not enough time in the day. So in order to comply with the new FDA regulations our Compounding Pharmacy in Texas had to reduce the number of my formulas from 100’s of single and various combinations down to only twelve.
We tried to make this work, but it was too limited, so in order to remedy the situation we moved our Compounding operations from Texas to Florida. We now have a Compounding Pharmacy in Florida who can make 36 of my most frequently prescribed single ingredient and combination Bioidentical Hormone formulas.
We realize that we are fighting against the current of the Mainstream Medicine and Big Drug Companies. They are trying to take away your options and your freedom to choose by preventing you from having options to choose. Mainstream Medicine and Big Drug Companies are also trying to squash the competition and put Compounding Pharmacies and Doctors who prescribe natural medicines out of business. Please don’t let that happen. We hope you will continue to fight with us by continuing to choose natural medicines and nutritional supplements because it is your right and because it is a safer option that will make you healthier.