The largest real-world human analysis to date evaluating ivermectin and mebendazole in cancer patients has just been published—and the results represent one of the most compelling clinical signals ever documented for repurposed anti-parasitic therapies in oncology.
This groundbreaking analysis was made possible through a unique collaboration between The Wellness Company, the McCullough Foundation, and the Chairman of the President’s Cancer Panel (Dr. Harvey Risch)—uniting real-world clinical data, frontline medical experience, and high-level epidemiologic expertise to deliver urgently needed insights in oncology.
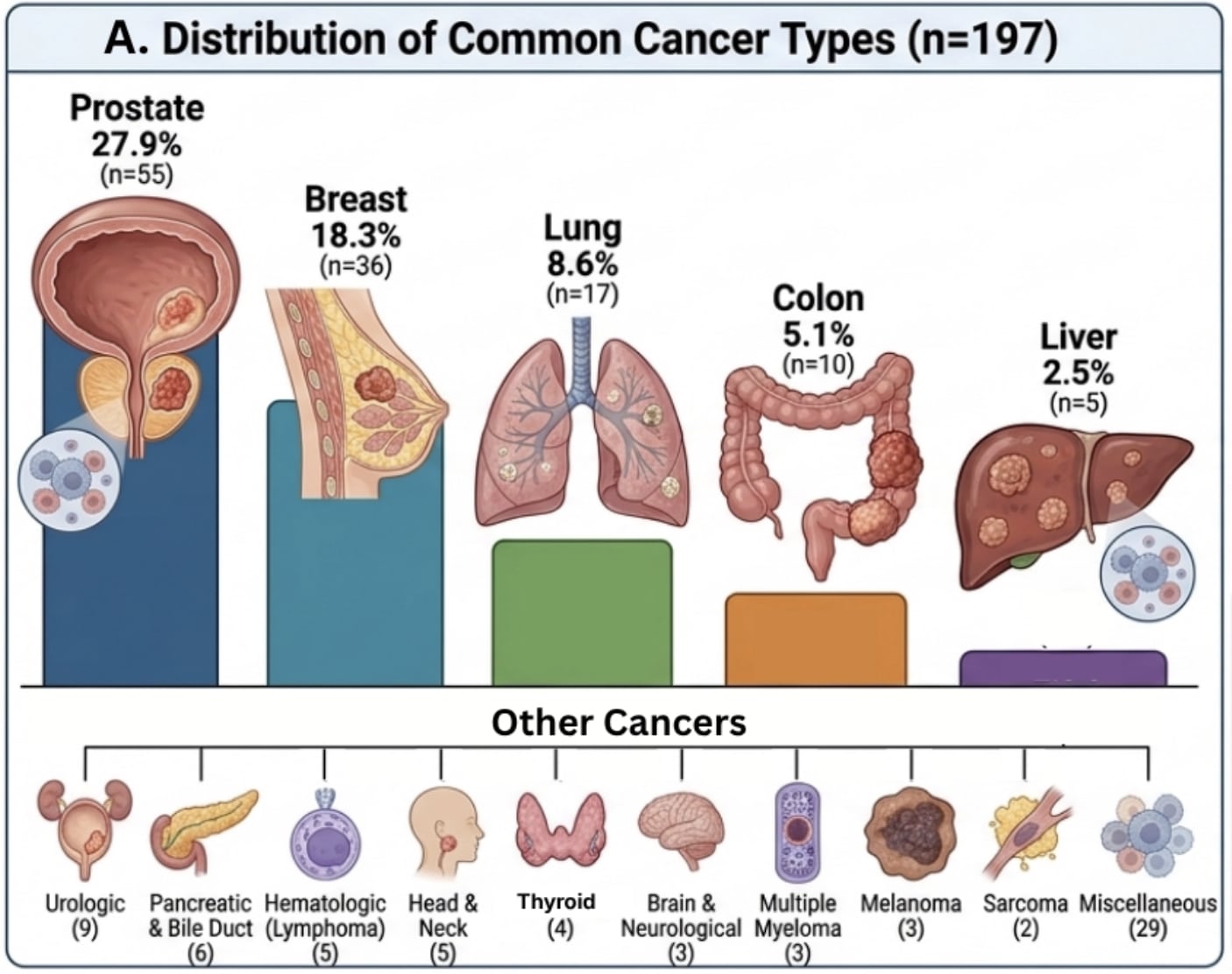
This was a real-world prospective clinical program evaluation of 197 cancer patients, with 122 completing a follow-up survey at about six months (61.9% response rate).
Cancer patients were prescribed compounded ivermectin–mebendazole, with each capsule containing 25 mg ivermectin and 250 mg mebendazole—most commonly taken at 1–2 capsules per day.
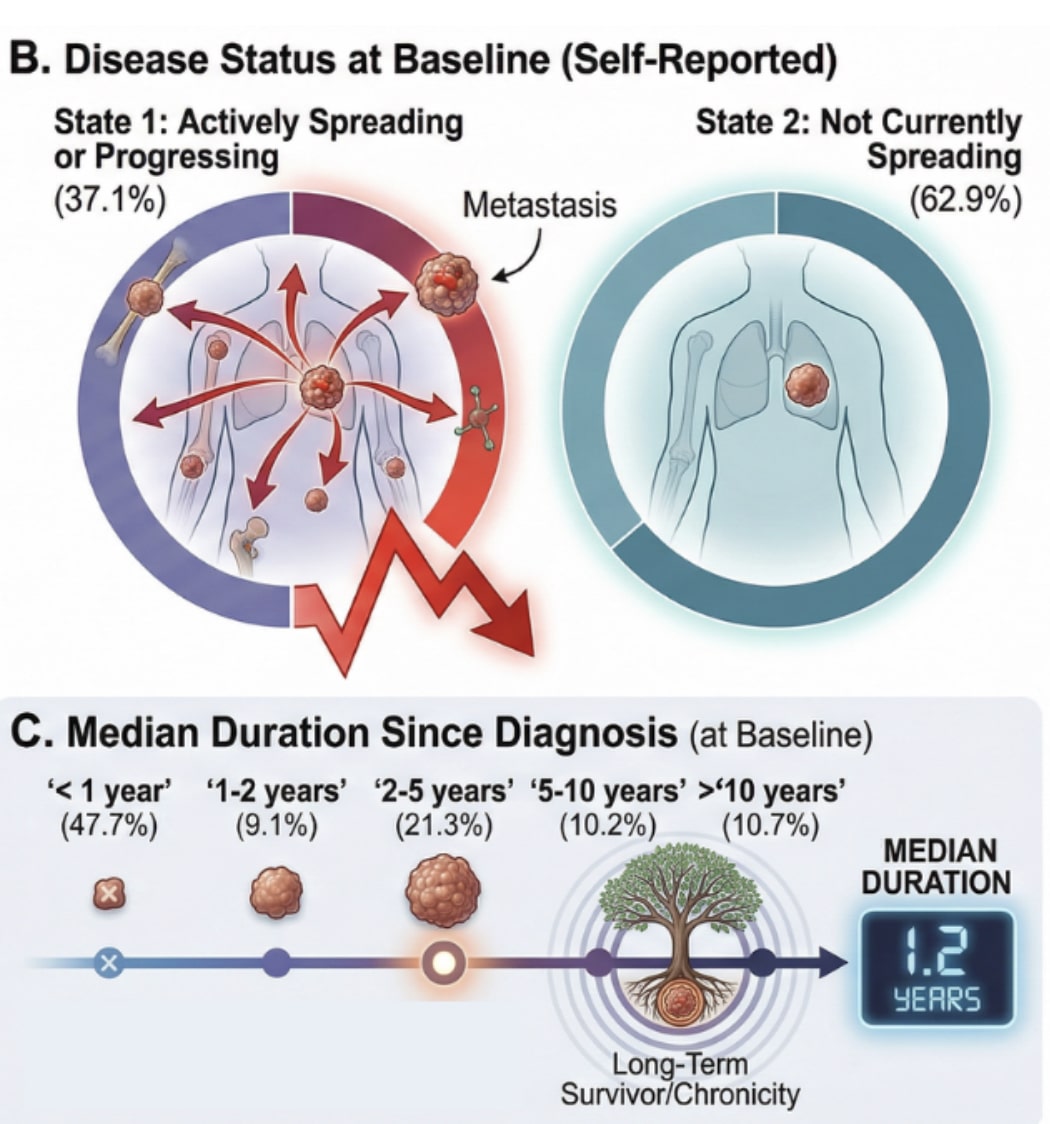
The cohort represented a clinically relevant population, including a wide variety cancer types, with 37.1% of patients reporting actively progressing disease at baseline and many having already undergone chemotherapy, radiation, and surgery.
At six months, 84.4% of cancer patients reported clinical benefit (Clinical Benefit Ratio: 84.4% [95% CI: 77.0–89.8%]):
✅ 32.8% reported NO evidence of cancer (95% CI: 25.1–41.5%)
✅ 15.6% reported tumor regression (95% CI: 10.2–23.0%)
✅ 36.1% reported stable disease (95% CI: 28.1–44.9%)
Treatment adherence was high, with 86.9% completing the full protocol and 66.4% remaining on therapy at six months.
The regimen was well tolerated, with 25.4% reporting side effects, primarily mild and gastrointestinal, and over 93% continuing treatment despite these events.
Patients were treated in real-world conditions alongside concurrent therapies, including chemotherapy (27.9%), radiation (21.3%), surgery (19.7%), supplements (49.2%), and dietary modification (37.7%), supporting use as an adjunctive approach.
Together, these findings represent a large, internally consistent real-world clinical signal that supports URGENT further investigation of ivermectin and mebendazole as low-toxicity, adjunctive cancer therapies.
Given the strength of the signal observed here, advancing this line of investigation is no longer optional—it is necessary.
This is NOT the end. We will continue advancing this work with larger datasets to further define and validate the role of anti-parasitics in cancer outcomes.
The manuscript is now available as a preprint on the Zenodo research repository, operated by the European Organization for Nuclear Research, while undergoing peer review at leading oncology journals: “Real-World Clinical Outcomes of Ivermectin and Mebendazole in Cancer Patients: Results from a Prospective Observational Cohort.”