A group of chemicals known as Pthalates have been found to increase the risk of cancer, diabetes, obesity. and now new research from Washington University, Missouri, indicates they may also cause early Menopause. Pthalates are found in plastics, cosmetics (including make-up and hairspray), household products and food packaging. Women exposed to higher levels have been found to have significant hormone fluctuations that cause the early onset of Menopause, an average of 2 1/2 years before other women.
Pthalates are believed to be one of the reasons so many women are experiencing prolonged Peri-Menoopaue symptoms, including hot flashes, moodiness, mental fogginess, sleeplessness, anxiety, depression, loss of libido, vaginal dryness, painful intercourse, weight gain and other symptoms of Menopause. In some cases, these chemicals may be causing women to stop having periods as much as 15 years too soon.
Dr Natalia Grindler, from Washington University in St Louis, Missouri, and colleagues looked at the levels of pthalates in the blood or urine of 5,700 women.
Pthalates have previously been linked to increased risks of cancer, diabetes and obesity. Now Dr. Grindler and other American researchers say the chemicals are disrupting women’s reproductive systems, including their ovaries, and leading to early menopause.
Those with the highest amounts were found to have gone through the menopause an average of 2.3 years before the others. The typical age of the menopause is 51, so women exposed to the highest levels were hitting it aged 49.
But Dr Grindler told the American Society of Reproductive Medicine’s conference in San Diego, California, that some women may be going through the menopause 15 years early, in their mid-thirties.
An early menopause is linked to far higher rates of strokes, heart disease, bone problems and fatal brain hemorrhages.
Dr Grindler said, ‘We don’t know yet if some of them are going through it one year earlier or some are going through it 15 years earlier.
‘Early menopause has a lot of impact on your health. We absolutely think these chemicals have the potential to affect ovarian function and human reproduction.
‘There’s a lot that we don’t know at this point, our research is still preliminary, but it’s enough to suggest it is having a detrimental impact in the long term.’
She could not explain why some women were exposed to higher levels of these chemicals. It may be that they wore more make-up, drank bottled water or ate more packaged foods.
‘Eating fresh, unpackaged food can reduce phthalate exposure but will not eliminate it.’
Chemical in Cosmetics Causing Early Menopause
Ivermectin + Mebendazole taken together produce remarkably Positive Clinical Cancer Benefits in 84.4% of Patients.
The largest real-world human analysis to date evaluating ivermectin and mebendazole in cancer patients has just been published—and the results represent one of the most compelling clinical signals ever documented for repurposed anti-parasitic therapies in oncology.
This groundbreaking analysis was made possible through a unique collaboration between The Wellness Company, the McCullough Foundation, and the Chairman of the President’s Cancer Panel (Dr. Harvey Risch)—uniting real-world clinical data, frontline medical experience, and high-level epidemiologic expertise to deliver urgently needed insights in oncology.
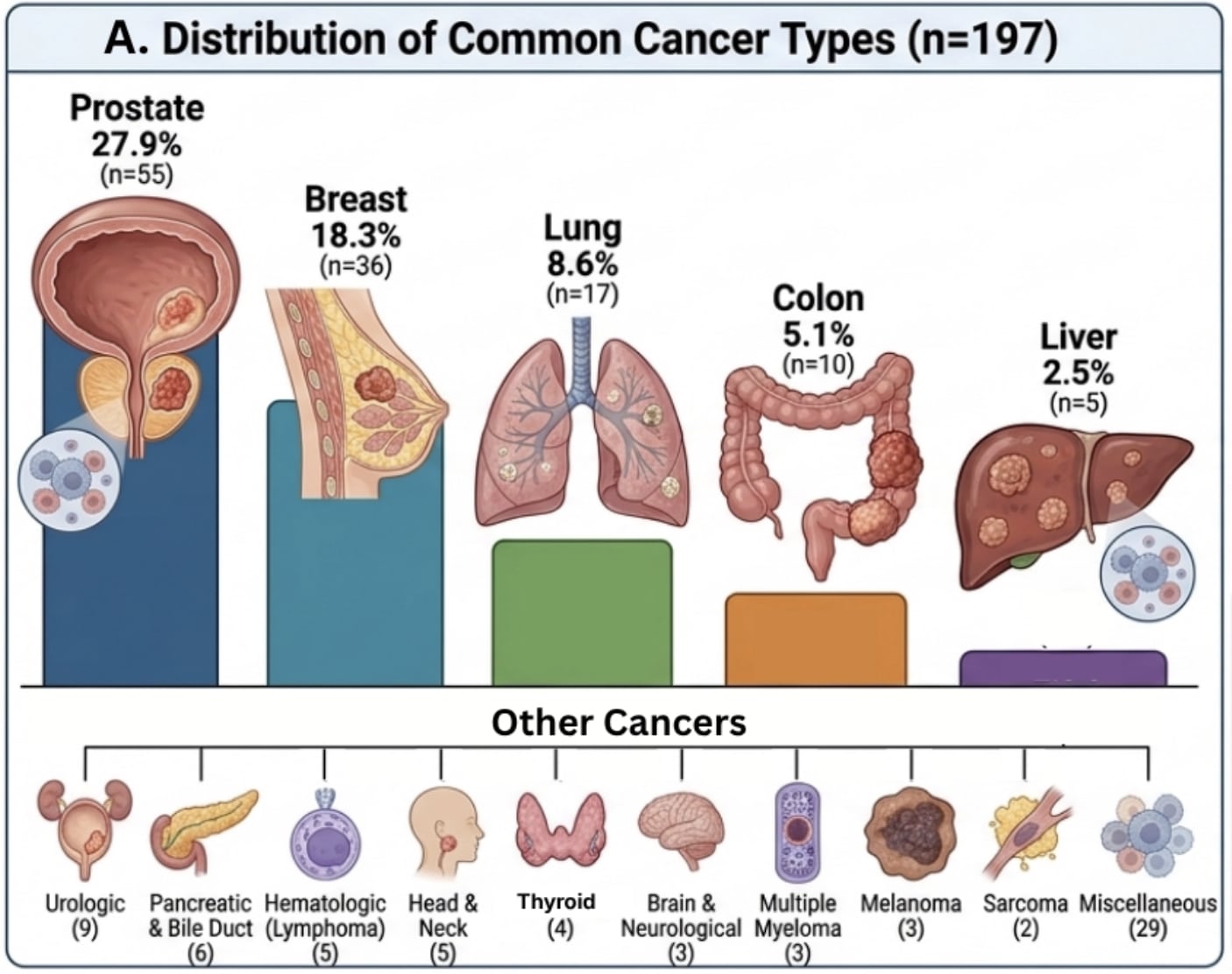
This was a real-world prospective clinical program evaluation of 197 cancer patients, with 122 completing a follow-up survey at about six months (61.9% response rate).
Cancer patients were prescribed compounded ivermectin–mebendazole, with each capsule containing 25 mg ivermectin and 250 mg mebendazole—most commonly taken at 1–2 capsules per day.
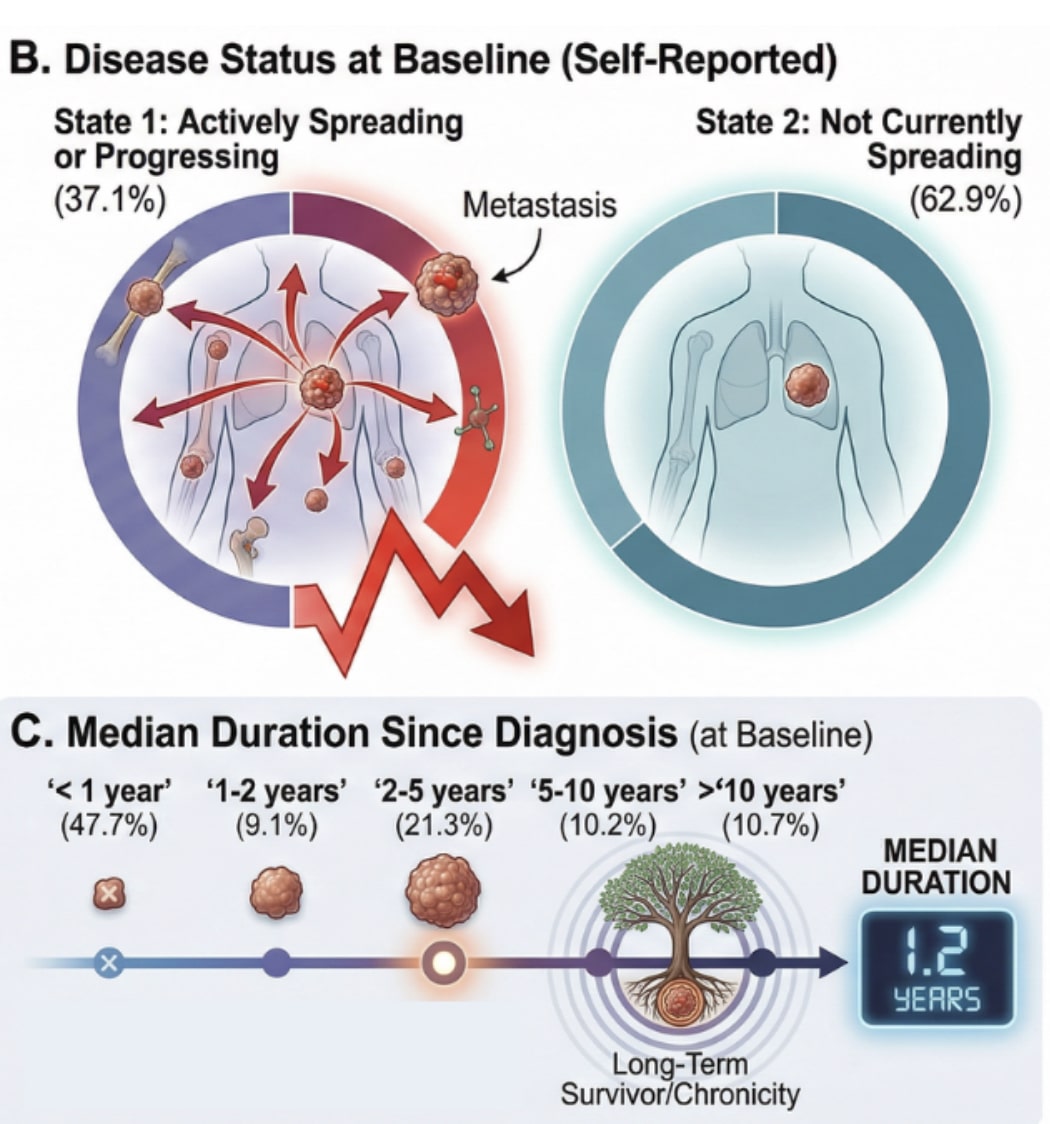
The cohort represented a clinically relevant population, including a wide variety cancer types, with 37.1% of patients reporting actively progressing disease at baseline and many having already undergone chemotherapy, radiation, and surgery.
At six months, 84.4% of cancer patients reported clinical benefit (Clinical Benefit Ratio: 84.4% [95% CI: 77.0–89.8%]):
✅ 32.8% reported NO evidence of cancer (95% CI: 25.1–41.5%)
✅ 15.6% reported tumor regression (95% CI: 10.2–23.0%)
✅ 36.1% reported stable disease (95% CI: 28.1–44.9%)
Treatment adherence was high, with 86.9% completing the full protocol and 66.4% remaining on therapy at six months.
The regimen was well tolerated, with 25.4% reporting side effects, primarily mild and gastrointestinal, and over 93% continuing treatment despite these events.
Patients were treated in real-world conditions alongside concurrent therapies, including chemotherapy (27.9%), radiation (21.3%), surgery (19.7%), supplements (49.2%), and dietary modification (37.7%), supporting use as an adjunctive approach.
Together, these findings represent a large, internally consistent real-world clinical signal that supports URGENT further investigation of ivermectin and mebendazole as low-toxicity, adjunctive cancer therapies.
Given the strength of the signal observed here, advancing this line of investigation is no longer optional—it is necessary.
This is NOT the end. We will continue advancing this work with larger datasets to further define and validate the role of anti-parasitics in cancer outcomes.
The manuscript is now available as a preprint on the Zenodo research repository, operated by the European Organization for Nuclear Research, while undergoing peer review at leading oncology journals: “Real-World Clinical Outcomes of Ivermectin and Mebendazole in Cancer Patients: Results from a Prospective Observational Cohort.”